|
|---|
CHRONO-PAR® Platelet Aggregation Reagents
Provide High Accuracy and Extremely Low Costs Per Test
|
Chrono-log offers a complete line of highly concentrated platelet aggregation reagents, requiring only micro-portions
per test. The standard aggregation reagents: Thrombin, ADP, Collagen, Ristocetin,
Epinephrine and Arachidonic Acid are available, as well as CHRONO-LUME® and ATP Standard reagents for determination of
dense granule release when performing Lumi Aggregation.
Please review this year-long study which confirmed the exceptional stability
of Chrono-log aggregation reagents.
CHRONO-PAR® Reagents are validated for use in both Whole Blood and PRP samples.
Click images below to enlarge. |
||
|
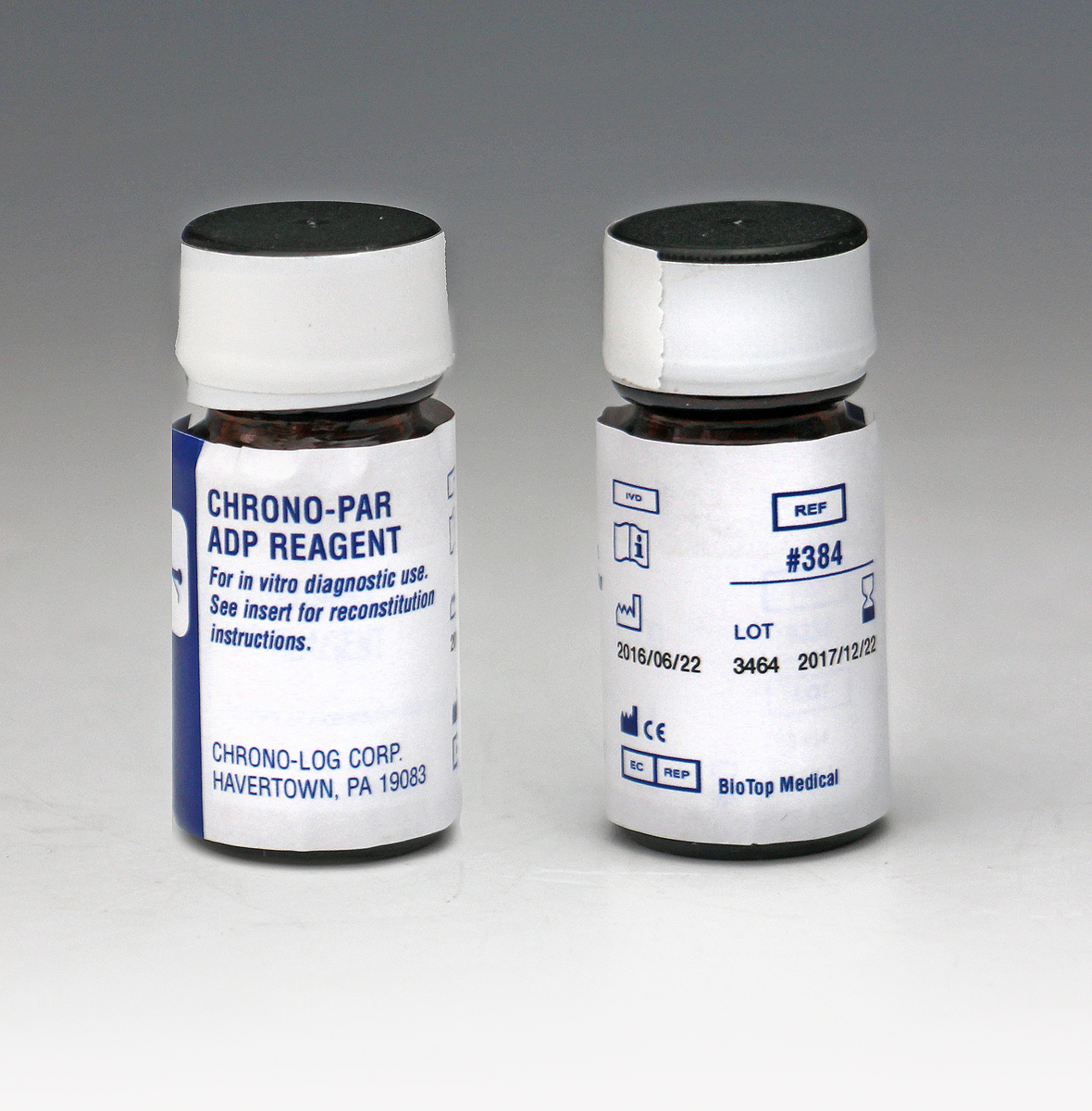
P/N 384 ADP Each vial contains 2.5mg of lyophilized adenosine diphosphate. Performs 500 to 1,000 tests in PRP or Whole Blood samples. |
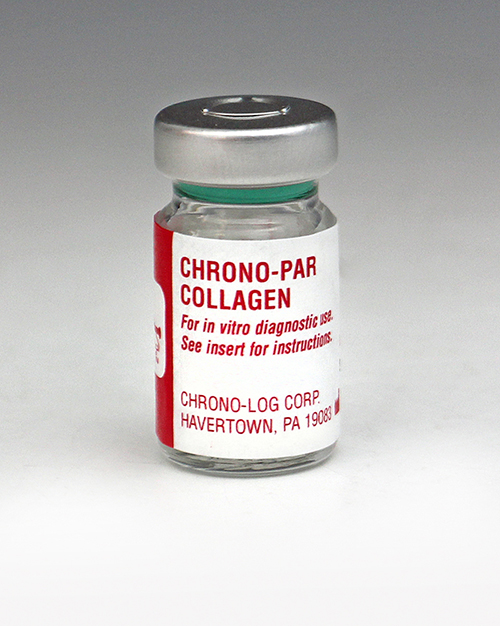
P/N 385 Collagen Each vial contains 1mg of native Collagen fibrils (type I) from equine tendons suspended in isotonic glucose solution of pH 2.7. Performs 500 to 1,000 tests in PRP or Whole Blood samples. |
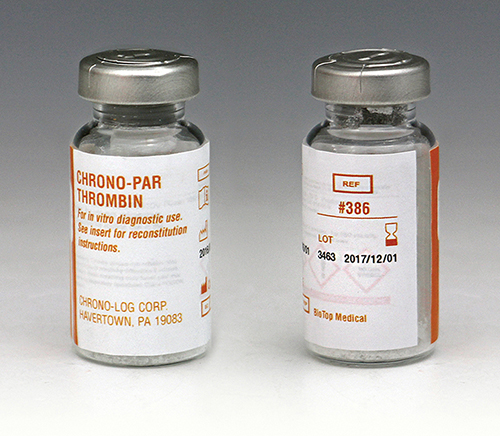
P/N 386 Thrombin Each vial contains a minimum of 10 units of lyophilized Thrombin from human plasma. Performs 10 to 20 tests in PRP or Whole Blood samples. |
|
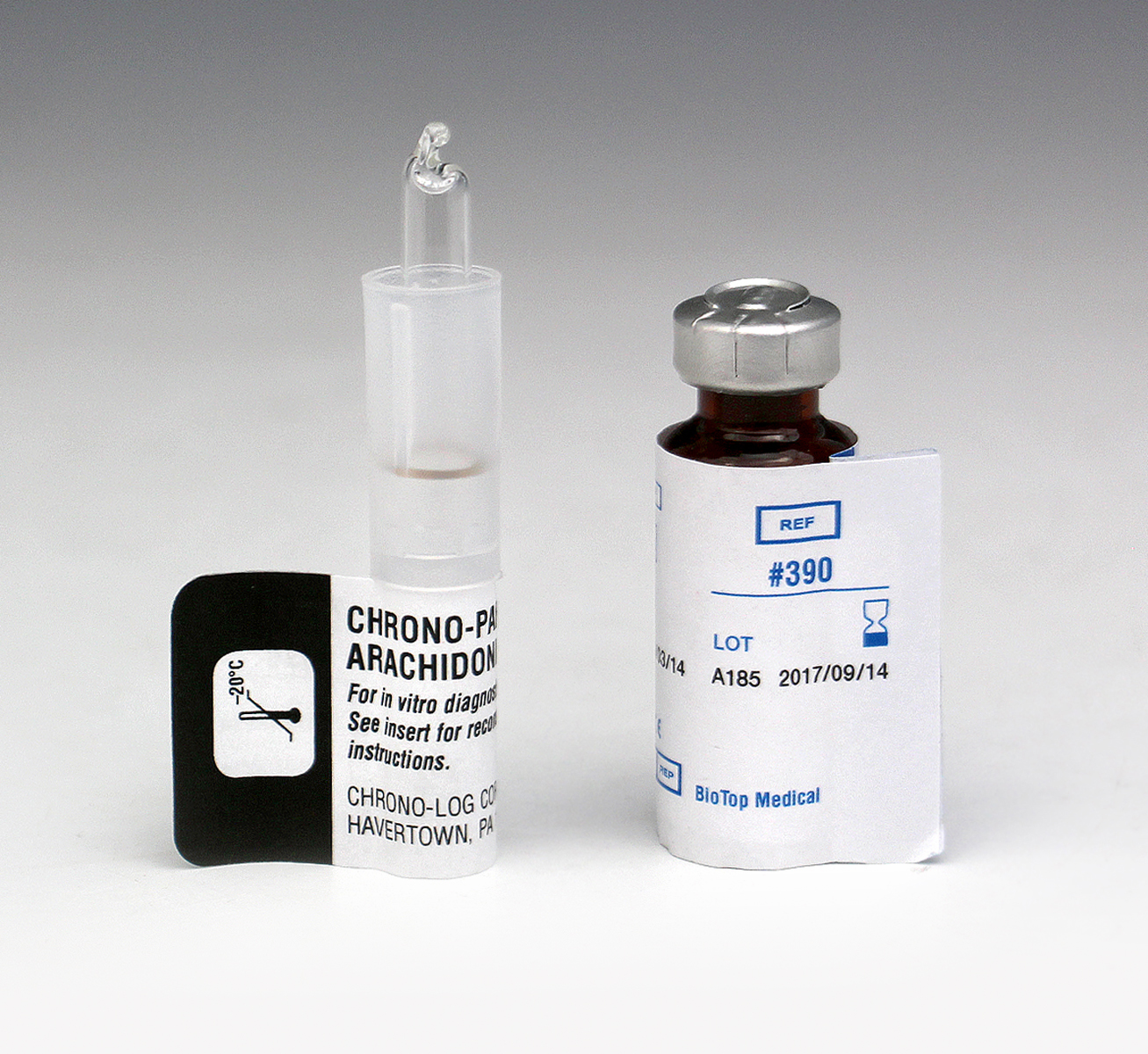
P/N 390
Arachidonic Acid Each vial contains a minimum of 10mg of Arachidonic Acid, purity better than 99%. Included is a vial containing 100mg bovine albumin, fraction V powder, 96% to 99% purity. Performs 70 to 140 tests in PRP or Whole Blood samples. |
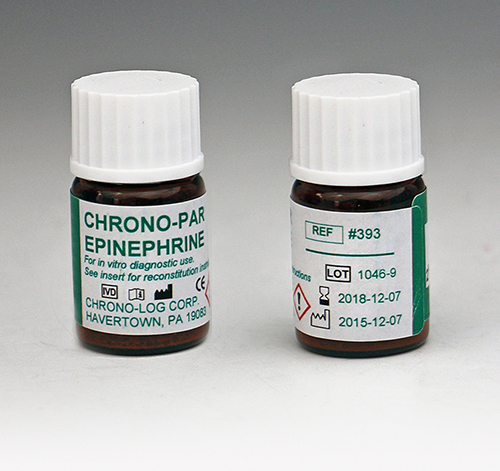
P/N 393
Epinephrine Lyophilized preparation of 1-Epinephrine bitartarate with stabilizers. Performs 1000 or more tests in PRP samples. |
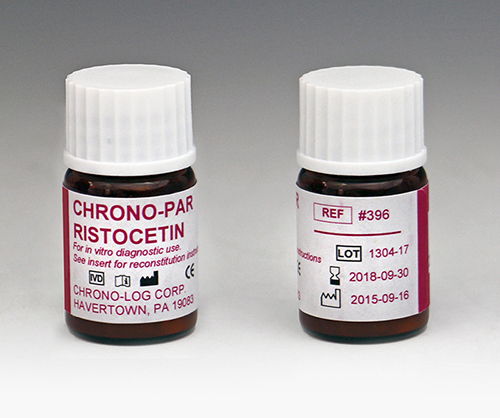
P/N 396
Ristocetin Each vial contains 62.5mg of stabilized freeze dried Ristocetin. Performs 62 to 125 tests in PRP or Whole Blood samples. |
CHRONO-LUME® Reagents
For Performing Secretion Study Simultaneously with Aggregation
|
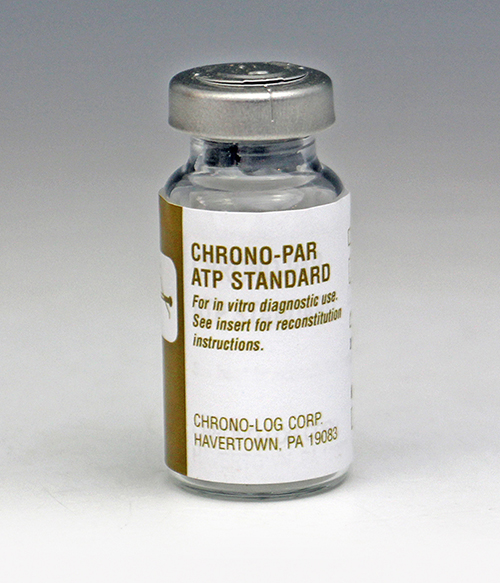
P/N 387
ATP Standard For the quantitation of ATP Release. Supplied as 2 μmole of lyophilized adenosine 5' triphosphate. 5 μL added to any size test sample provides a 2 nmole standard. |
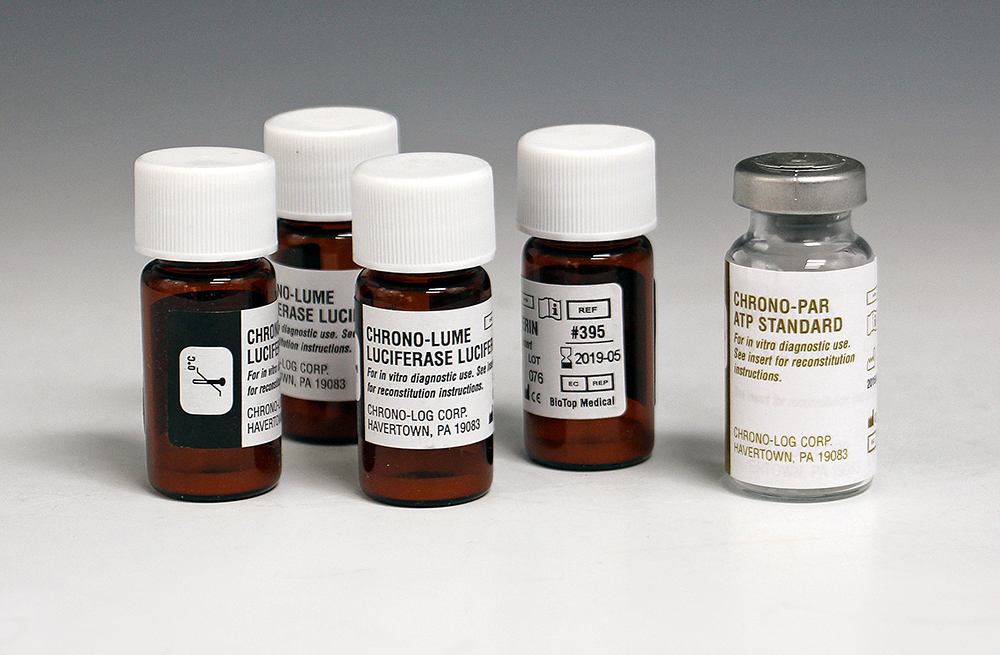
P/N 395
CHRONO-LUME® Reagent for measurement of ATP release. Each vial contains 0.2 mg luciferin, 22,000 units d-luciferase plus magnesium sulfate, human serum albumin, stabilizers and buffer. Kit includes 4 vials of CHRONO- LUME plus a vial of lyophilized adenosine 5' triphosphate for use as an ATP Standard. Performs 50 to 100 tests in PRP or Whole Blood samples. |
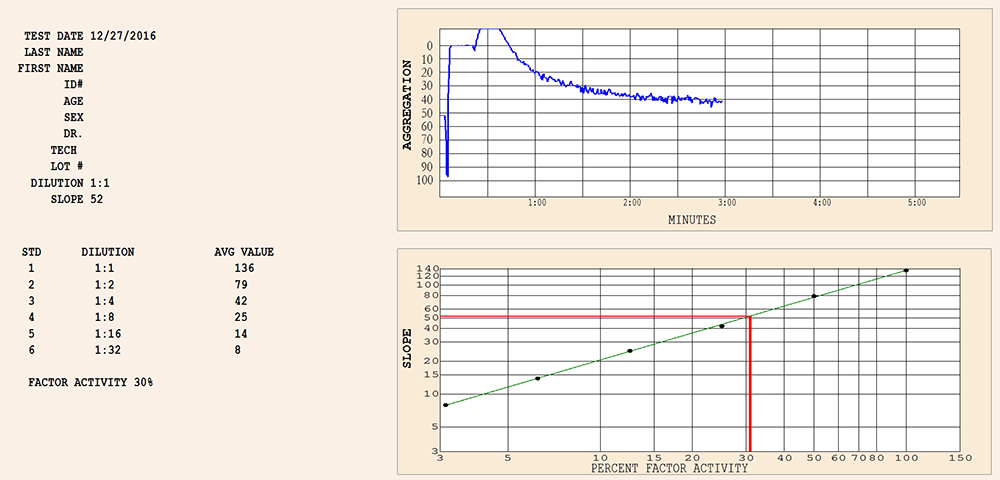
Ristocetin CoFactor Assay
For the Quantitation of Ristocetin CoFactor Activity
|
P/N 299
Ristocetin CoFactor Assay Kit (von Willebrand Factor Assay)
For use in the quantitative determination of Factor VIII Ristocetin Cofactor Activity in citrated plasma. The following Materials included provide volumes sufficient to run 15 determinations: Ristocetin, |
P/N 299-1
Ristocetin, 7.5mg/Vial
For use in the quantitative determination of Factor VIII Ristocetin Cofactor Activity in citrated plasma. Each vial contains 7.5 mg of |
P/N 299-2
Lyophilized Human Platelets 6mL, Tris Buffered Saline 12mL
For use in the quantitative determination of Factor VIII Ristocetin Cofactor Activity in citrated plasma. Each vial provides 6 mL of Platelets following reconstitution with |
|
P/N 299-3
vW Reference Plasma Normal 1mL
For use in the quantitative determination of Factor VIII Ristocetin Cofactor Activity in citrated plasma. Each vial contains dried human plasma |
P/N 299-4
vW Reference Plasma Deficient .5mL
For use in the quantitative determination of Factor VIII Ristocetin Cofactor Activity in citrated plasma. Each vial contains |
P/N 299-5
Tris Buffered Saline 12m
For use in the reconstitution of lyophilized Platelets, used in the quantitative determination of Factor VIII Ristocetin |
|
P/N 299-6 Hemostasis Reference Plasma 1mL
For use as a Reference Plasma in the quantitative determination of Factor VIII Ristocetin Cofactor Activity in citrated plasma. Hemostasis Reference Plasma Abnormal 1mL
For use as an Abnormal Reference Plasma in the quantitative determination of Factor VIII Ristocetin Cofactor Activity in citrated plasma. |
|
Chrono-log also provides a complete system to perform the RISTOCETIN COFACTOR ASSAY including:
|
![]()